
How to Store Peptides After Reconstitution Without Losing Potency
Knowing how to store reconstituted peptides correctly is critical for

A Comprehensive Clinical Reference for Skincare Professionals and Estheticians
Neuropeptides have reshaped the cosmeceutical landscape. Over the past two decades, they have become one of the most credible non-invasive tools for reducing dynamic facial wrinkles.
SNAP-8 sits at the top of this peptide category. Its formal INCI designation is Acetyl Glutamyl Heptapeptide-1. It is also referred to as Acetyl Octapeptide-3. Both names describe the same synthetic octapeptide engineered specifically to disrupt the neuromuscular pathway that creates expression lines.
For skincare professionals and estheticians, understanding SNAP-8 Peptide is not optional. It is a clinical necessity. This article covers everything: molecular structure, mechanism of action, clinical evidence, formulation science, and application protocols. It is built for practitioners who need precision, not marketing language.
SNAP-8 is a synthetic octapeptide. It consists of a chain of eight amino acids.
It was developed by Lipotec S.A., now part of Lubrizol Life Science. The compound was designed as a structural extension of Argireline, the Acetyl Hexapeptide-3 that preceded it. Argireline mimics the N-terminal segment of SNAP-25, a key protein in neurotransmitter release. SNAP-8 takes that concept further. Two additional amino acids were added to the chain. The result is a more stable molecule with a stronger binding geometry at the SNARE complex interface.
| Key Identity Data INCI Name: Acetyl Glutamyl Heptapeptide-1 | Also Known As: Acetyl Octapeptide-3 | Chain Length: 8 Amino Acids | Developed By: Lipotec S.A. (Lubrizol) | Parent Compound: Argireline (Acetyl Hexapeptide-3) |
Dynamic wrinkles are not simply a surface problem. They originate at the neuromuscular junction.
Every facial expression triggers muscle contraction. That contraction is driven by acetylcholine, a neurotransmitter released at the synapse. The release process is controlled by a protein assembly called the SNARE complex.
Three proteins form the core of this complex:
When these three proteins assemble, they fuse the vesicle to the membrane. Acetylcholine is released. The muscle contracts. Over time, repetitive movement permanently deforms dermal collagen. Expression lines become fixed.
That is the problem SNAP-8 is designed to address.
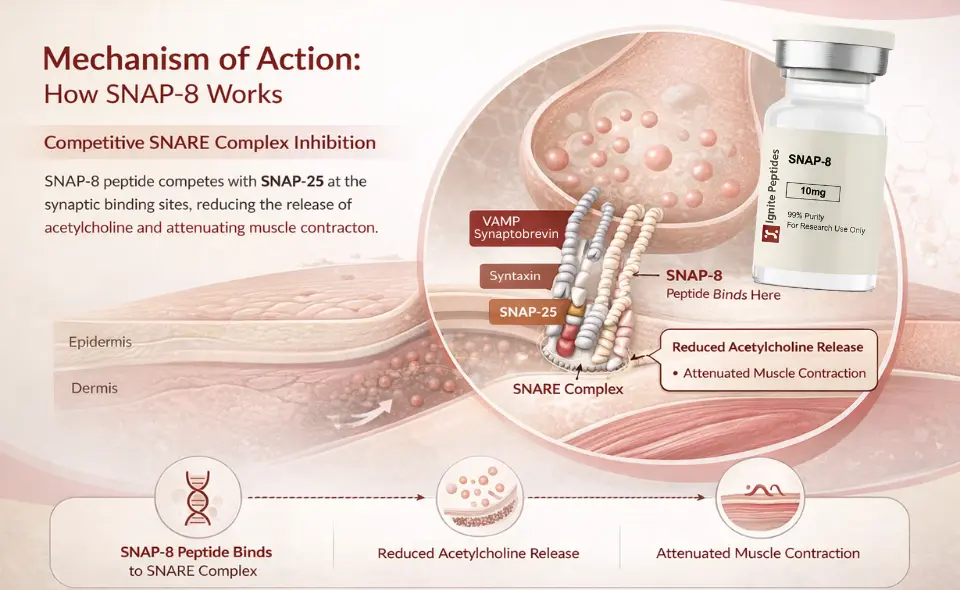
SNAP-8 does not block nerves. It does not destroy proteins. It competes.
The peptide mimics the N-terminal domain of SNAP-25. It presents a structurally similar sequence to the target binding site within the assembling SNARE complex. When SNAP-8 occupies those binding positions, the ternary complex cannot fully form.
The consequence is direct. Synaptic vesicles cannot fuse efficiently with the presynaptic membrane. Neurotransmitter release is reduced. Muscle contraction is attenuated. Over time, consistent use softens dynamic expression lines.
| Clinical Implication SNAP-8 peptide works through competitive inhibition, not enzymatic cleavage. Its effects are dose- and concentration-dependent and fully reversible. This means natural facial movement is preserved. There is no freezing. The risk profile is fundamentally different from injectable neuromodulators. |
Clients frequently ask how SNAP-8 compares to Botox. The comparison is clinically relevant but only when framed accurately. These two modalities share a target but operate through completely different mechanisms.
| Parameter | SNAP-8 (Acetyl Octapeptide-3) | Botulinum Toxin Type A |
| Mechanism | Competitive inhibition of the SNARE complex | Enzymatic cleavage of SNAP-25 protein |
| Effect on the muscle | Gradual, partial attenuation of contraction | Near-complete, temporary paralysis |
| Route of delivery | Topical (epicutaneous) | Intramuscular injection |
| Reversibility | Fully reversible; requires ongoing use | Reversible over 3-6 months (nerve regeneration) |
| Onset of visible results | 4-8 weeks of consistent twice-daily use | 3-7 days post-injection |
| Risk profile | Very low; generally well tolerated | Bruising, asymmetry, ptosis, systemic risks |
| Expression preservation | Natural movement preserved | Movement may be restricted or frozen |
| Practitioner requirement | None (for topical formulations) | Licensed injector required |
| Regulatory classification | Cosmetic ingredient | Prescription drug (Class II medical device) |
SNAP-8 peptide is not a Botox replacement. That framing does a disservice to both modalities. SNAP-8 occupies a distinct clinical niche. It is the tool of choice for preventive anti-aging, maintenance between injection cycles, and as a primary non-invasive option for clients who decline injectables.
Lipotec’s original validation used an in vitro assay. The assay evaluated the ability of small peptides to compete with native SNAP-25 during reconstitution of the SNARE complex.
The findings were clear. SNAP-8 demonstrated approximately 30% greater inhibitory activity than Argireline. The two additional amino acids in SNAP-8’s chain are responsible. They improve conformational stability and binding geometry at the SNARE interface.
The landmark in vivo study enrolled 17 healthy female volunteers. The protocol was straightforward: twice-daily application of a 10% SNAP-8 cream for 28 days. Silicon imprints of the periorbital area were captured at baseline and endpoint. Three-dimensional confocal laser scanning microscopy was used to assess changes in wrinkle depth.
| Key Clinical Finding Maximum periorbital wrinkle depth reduction: 63.13%. Mean reduction: approximately 34.98%. The 10% Argireline control group achieved a -27.05% change. The placebo group showed a -2.99% change. These results position SNAP-8 as the stronger performer among SNARE-targeting peptides at equivalent concentrations. |
A second trial, published in the Annals of Dermatology, examined SNAP-8 delivered via a dissolving microneedle patch. The study ran for four weeks across 24 subjects. Results showed significant improvement in both wrinkle depth and skin elasticity. No adverse effects were reported.
A third assessment, published in the Journal of Drugs in Dermatology, evaluated a multi-peptide serum containing SNAP-8 over 14 weeks. The open-label trial reported statistically significant expression line smoothing. Tolerability was favorable across all participants.
The evidence is promising. But it requires context.
Most published SNAP-8 efficacy data originate from manufacturer-sponsored studies. Independent academic research is more limited. It tends to report conservative outcomes, typically 10-30% wrinkle depth reduction under real-world conditions. The 63% figure comes from an optimized clinical trial setting.
Several factors influence real-world results: formulation quality, delivery vehicle, concentration, application consistency, individual neuromuscular anatomy, and client compliance.
SNAP-8 delivers measurable, progressive improvement with consistent use. It is not a surrogate for injectable neuromodulators in clients presenting with established moderate-to-severe rhytides. Set expectations accordingly.
SNAP-8 is commercially supplied as a pre-dissolved solution. The standardized active concentration is 0.05% Acetyl Octapeptide-3 in an aqueous base, typically preserved with Caprylyl Glycol. This solution is then incorporated into finished formulations at defined usage rates.
| Formulation Concentration | Target Indication | Clinical Notes |
| 3% SNAP-8 solution | Sensitive or reactive skin; introductory use | Lower irritation risk; well-suited to delicate periorbital skin |
| 5-7% SNAP-8 solution | Maintenance protocols; combination anti-aging regimens | Balanced efficacy and tolerability for most skin types |
| 10% SNAP-8 solution | Maximum anti-wrinkle effect; professional clinical use | Clinically validated; optimal for periorbital, frontal, and glabellar rhytides |
The 10% concentration is the benchmark. It is the level established in principal efficacy studies and recommended for professional-grade anti-aging formulations.
For client home-care dispensing, begin at 3-5%. Progress to 10% once tolerance is confirmed.
SNAP-8 peptide is water-soluble. It must be incorporated into aqueous-phase formulations. Anhydrous and oil-only bases are not compatible.
Suitable vehicles include:
The temperature rule is critical. SNAP-8 must be added after the formulation cools below 40°C. Heat degrades peptide integrity. This is non-negotiable in any professional compounding setting.
| Formulation Stability Warning Do not incorporate SNAP-8 into strongly acidic formulations (pH below 3.5). Avoid direct combination with high-concentration ascorbic acid at low pH. Both conditions destabilize the peptide backbone and reduce bioactivity. Maintain formulation pH between 5.0 and 7.0 for optimal stability. |
Store raw SNAP-8 solution and all finished formulations at 4°C (39°F). Refrigeration preserves peptide stability.
SNAP-8 is light-sensitive. Use UV-protective packaging, such as amber glass or opaque containers, for both clinical stock and client-dispensed products.
One known artefact: refrigerated SNAP-8 peptide solution may cause the Caprylyl Glycol preservative to precipitate. This does not affect peptide integrity. It resolves with gentle warming. Advise clients of this when dispensing home-care products.
Correct application directly determines how much of SNAP-8’s potential is realized. The peptide needs adequate concentration at the target tissue interface to produce competitive inhibition. Technique matters.
SNAP-8 is most effective where dynamic contraction-driven rhytides dominate. These zones are the primary targets for treatment planning:
| Target Zone | Primary Muscle | Expression Pattern Addressed |
| Frontalis region (horizontal forehead lines) | Frontalis m. | Brow elevation, surprise, and concentration lines |
| Glabellar complex (vertical frown lines / ’11s’) | Corrugator supercilii, Procerus | Medial brow furrowing; anger and concentration lines |
| Periorbital zone (crow’s feet) | Orbicularis oculi (lateral) | Lateral canthal rhytides from smiling and squinting |
| Upper lip perioral lines | Orbicularis oris | Repetitive lip movement; smoker’s lines |
| Marionette lines | Depressor anguli oris | Downward mouth corner pull from repeated expression |
Position SNAP-8 serum correctly within the layering sequence. Incorrect placement risks bioactivity loss and reduced penetration.
Step 1 Cleanse: Clean the area with a mild, pH-balanced cleanser. Avoid strongly acidic or surfactant-heavy formulas that disrupt the epidermal barrier.
Step 2 Tone / Prep (Optional): Apply a hydrating mist or toner. Avoid alcohol-based astringents.
Step 3 SNAP-8 Serum: Apply directly to target zones. Wait about 60–90 seconds for it to fully absorb before moving on to the next step.
Step 4 Supporting Actives (Optional): Apply complementary peptides such as Matrixyl 3000 or GHK-Cu, followed by hyaluronic acid serum or growth factor preparations.
Step 5 Moisturizer / Barrier Support: Seal with a ceramide-rich or occlusive moisturizer to protect the integrity of the stratum corneum.
Step 6 SPF (AM only): Broad-spectrum SPF 30 or higher is mandatory in the morning routine. UV exposure accelerates SNAP-25 degradation and undermines every anti-aging investment.
The SNAP-8 peptide has an excellent safety profile. Adverse reactions are rare. When they occur, they are typically mild and transient, usually localized erythema in hypersensitive individuals.
Apply the following professional precautions:
SNAP-8 peptide pairs well with a range of anti-aging actives. Strategic multi-ingredient protocols address several aging mechanisms simultaneously. Single-peptide approaches rarely match the depth of improvement achievable through evidence-informed stacking.
| Partner Ingredient | Mechanism | Clinical Synergy with SNAP-8 |
| Matrixyl 3000 (Palmitoyl Tripeptide-1 + Palmitoyl Tetrapeptide-7) | Stimulates collagen I, III, IV; reduces IL-6 inflammatory signalling | Targets structural dermal ageing collagen depletion, while SNAP-8 addresses the neuromuscular driver of expression lines |
| GHK-Cu (Copper Peptide) | Promotes collagen and elastin synthesis; wound healing; antioxidant defence | Adds a regenerative layer; especially valuable in post-procedure integration protocols |
| Argireline (Acetyl Hexapeptide-3) | SNARE complex inhibition via SNAP-25 N-terminal mimicry | Additive neuromuscular inhibitory effect; combining with Leuphasyl produces stronger glutamate pathway inhibition |
| Leuphasyl (Pentapeptide-18) | Enkephalin receptor agonist; reduces glutamate-driven muscle activation | Dual-pathway neuromodulation SNARE complex inhibition plus enkephalin receptor engagement measurably amplifies muscle relaxation |
| Hyaluronic Acid (multi-molecular weight) | Hygroscopic humectant; deep and surface hydration | Optimizes skin hydration to volumize fine lines; serves as an ideal aqueous delivery vehicle for the peptide |
| Retinoids (Retinol, Retinal, Tretinoin) | Upregulates collagen synthesis; increases cell turnover; reverses photoaging | The gold-standard combination: retinoids address photoaging and collagen loss while SNAP-8 targets the neuromuscular component of wrinkle formation |
Device-assisted delivery amplifies SNAP-8’s clinical impact. The rate-limiting barrier is the stratum corneum. The following technologies overcome it.
Microneedling (Collagen Induction Therapy): Needling creates temporary micro-channels in the epidermis. These channels allow SNAP-8 to penetrate the papillary and reticular dermis, well beyond the reach of passive topical application. A 24-subject clinical trial confirmed significantly greater wrinkle reduction with microneedle patch delivery versus standard topical application.
Iontophoresis: Direct electrical current drives charged peptide molecules electrophoretically across the epidermis. SNAP-8’s molecular charge profile makes it a suitable candidate. Confirm formulation compatibility with the iontophoretic device before use.
Sonophoresis (Ultrasound-Assisted Delivery): Low-frequency ultrasound temporarily disrupts the packing of lipid bilayers in the stratum corneum. This creates an acoustic absorption pathway for macromolecular ingredients. Professional ultrasound devices at 1-3 MHz are standard in esthetic clinical practice.
SNAP-8 and Argireline share a mechanism and a structural lineage. In practice, practitioners must choose between them or strategically combine them. Use this framework to guide that decision.
| Clinical Criterion | Preferred Choice | Rationale |
| Maximum anti-wrinkle efficacy required | SNAP-8 | Approximately 30% more active than Argireline in vitro; superior wrinkle depth reduction at 10% concentration |
| Cost-sensitive formulation | Argireline | More economical per unit weight; suitable for retail and home-care product ranges |
| Highly sensitive or reactive skin | Argireline or SNAP-8 at 3% | A shorter peptide chain may be marginally better tolerated in hypersensitive presentations |
| Maximum glutamate pathway inhibition | SNAP-8 + Leuphasyl combination | Dual-pathway inhibition significantly amplifies muscle relaxation over any single-peptide protocol |
| Maintenance between BoNT-A injection cycles | SNAP-8 at 10% | Sustains partial neuromuscular modulation during the regression phase of injectable treatment |
| Professional compounding or custom serum | SNAP-8 at 10% in HA base | Well-characterized formulation; straightforward incorporation into aqueous vehicles |
Setting accurate expectations is one of the most important clinical skills when implementing SNAP-8 peptide protocols. The gap between marketing claims and clinical reality is wide. Bridging it with evidence-based communication protects client trust and professional credibility.
| Timeframe | Expected Observable Change |
| Weeks 1-2 | Subtle improvements in surface texture and hydration. No significant wrinkle reduction yet. |
| Weeks 3-4 | Early softening of fine lines in high-expression zones (periorbital, frontal) with consistent twice-daily use. |
| Weeks 5-8 | Measurable reduction in wrinkle depth. Dynamic lines appear less defined at rest. |
| Weeks 8-12+ | Results plateau at the optimal level. Maximum neuromuscular modulation achieved. Maintenance use is required to sustain outcomes. |
| Post-discontinuation | Effects reverse within days to weeks. Native SNAP-25 resumes full participation in the SNARE complex upon competitive displacement. |
Clients with moderate-to-severe established dynamic rhytides need honest guidance. SNAP-8 produces clinically meaningful results. But it will not replicate the magnitude or speed of botulinum toxin injection.
Frame SNAP-8 accurately: it is a non-invasive adjunct, a maintenance tool, and a preventive strategy. It is not a direct injectable substitute for advanced presentations.
For clients who firmly decline injectables, document realistic expectations clearly in the consultation record. This protects both the client relationship and professional accountability.
No single ingredient resolves the full spectrum of skin aging. SNAP-8 excels in one specific domain: the neuromuscular component of dynamic wrinkle formation. Its role is most powerful when embedded in a multimodal treatment architecture.
A complete anti-aging protocol addresses five distinct aging mechanisms simultaneously:
Within this architecture, SNAP-8 is positioned as a daily home-care active and a professional treatment booster. It complements device-assisted procedures and injectables. It does not replace them.
Its non-invasive nature, strong tolerability profile, and growing evidence base make it one of the most valuable neuropeptide options available to the contemporary esthetician.
SNAP-8 peptide is a scientifically grounded anti-ageing ingredient with a clearly defined molecular target and a growing clinical evidence base.
Its value is not in bold marketing comparisons. It is in precision. SNAP-8 topically delivers measurable neuromuscular modulation, reducing the muscle-contraction component of dynamic wrinkle formation without the risks and restrictions of injectable neuromodulators.
For optimal outcomes, use a 10% concentration in an aqueous vehicle. Apply twice daily for at least 8 weeks. Combine with collagen-stimulating peptides such as Matrixyl 3000. Where clinically appropriate, pair with device-assisted delivery to enhance transdermal penetration.
For the skincare professional who demands evidence-based practice, SNAP-8 represents a mature, well-characterized tool. Use it correctly, position it accurately, and it delivers results that justify its place at the top of the non-invasive anti-aging peptide category.

Knowing how to store reconstituted peptides correctly is critical for

Retatrutide is a triple hormone receptor agonist. It works by

For decades, metabolic pharmacology focused on one goal: reducing calorie
Several factors shape retatrutide dosage in clinical research. These include

At Ignite Peptides, we provide a secure and reliable way to source high-quality research peptides online. As a trusted U.S.-based peptide supplier, we focus on lab-verified products, full transparency, and consistent quality. With fast shipping and responsive support, we’re committed to advancing safe, dependable research you can trust.
This helps us maintain strict standards for regulatory compliance, researcher verification, and responsible product access.