How to Calculate mg to mL for Peptide Research (Step-by-Step)
Every peptide vial in a research laboratory is labelled in

For decades, metabolic pharmacology focused on one goal: reducing calorie intake. GLP-1 compounds first proved this idea. Dual agonists refined it further.
However, energy expenditure remained mostly unchanged. This continued until retatrutide was introduced. Retatrutide is a 39-amino acid peptide. It adds glucagon receptor activity. This opened a completely new pathway in metabolic research.
Retatrutide is an investigational compound developed by Eli Lilly and Company. Its development code name is LY3437943. In scientific literature, it is referred to as a triple-G compound because it targets three hormone receptors simultaneously.
Most metabolic compounds work on one or two hormone pathways. Retatrutide goes further. It activates the GLP-1, GIP (glucose-dependent insulinotropic polypeptide), and glucagon (GCG) receptors. This makes it a triple-hormone receptor agonist, meaning it mimics three natural hormones at once.
To understand how retatrutide works, it is essential to understand the three hormones it mimics. Each hormone has a distinct physiological function.
GLP-1 is a natural hormone released from the gut after food intake. It forms the biochemical basis of modern metabolic drug research. When the GLP-1 receptor is activated, three key processes occur:
Together, these effects reduce caloric intake and stabilise post-meal blood glucose levels.
GIP is an incretin hormone released from the gut after eating. It works alongside GLP-1 in regulating metabolic responses. When the GIP receptor is activated, several important processes follow:
A critical biochemical detail: retatrutide is 8.9 times more potent at the GIP receptor than native GIP itself. Its EC50 value at the GIP receptor is 0.0643 nM. This exceptional potency at the GIP receptor is a key contributor to the compound’s strong metabolic profile.
This is where retatrutide truly stands apart from all other metabolic compounds currently in research. Glucagon is a natural hormone produced in the pancreas, classically known for raising blood glucose. Targeting it in a metabolic compound might initially seem counterintuitive.
However, glucagon receptor (GCGR) activation at the doses used in retatrutide research produces a powerful and unique set of effects:
The simultaneous activation of GLP-1 and GIP receptors neutralises glucagon’s hyperglycaemic risk. Together, these three pathways maintain glucose balance while still delivering the fat-oxidation benefits of glucagon receptor activation.
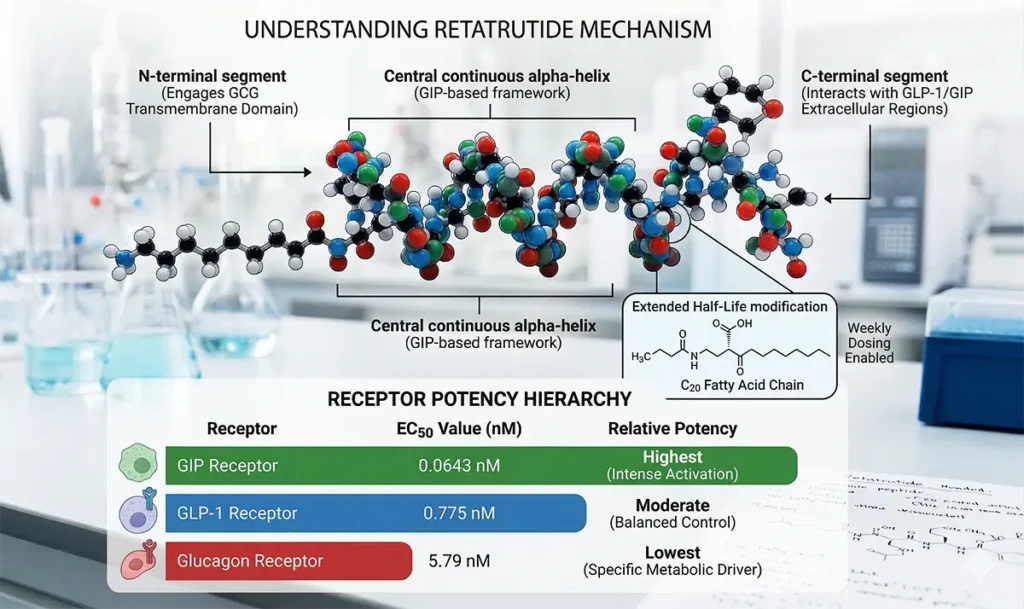
Retatrutide is a 39-amino acid peptide. Researchers designed the molecule using a structural framework based on the GIP peptide. The molecule adopts a single continuous helical structure. This design allows it to bind to all three receptors using different segments of the peptide chain.
This is why a single molecule can activate three different receptor systems.
To extend its biological activity, researchers linked retatrutide to a C20 fatty acid. This modification is central to its long half-life. Retatrutide remains biologically active for approximately six days, enabling once-weekly dosing intervals.
The compound undergoes hepatic metabolism and does not interact with CYP450 enzymes, reducing the potential for compound interactions.
Retatrutide’s potency across its three receptors follows a deliberate hierarchy:
| Receptor | EC50 Value | Relative Potency |
| GIP Receptor | 0.0643 nM | Highest |
| GLP-1 Receptor | 0.775 nM | Moderate |
| Glucagon Receptor | 5.79 nM | Lowest |
This profile is intentional. It balances powerful metabolic activation with manageable glucagon receptor effects.
The true power of the retatrutide mechanism lies in the combination of all three receptor pathways. Each receptor adds a distinct layer of metabolic activity. When all three operate simultaneously, the result is greater than the sum of its parts.
The mechanism operates on two simultaneous metabolic fronts:
Front 1 — Reduce Intake: GLP-1 and GIP receptor activation suppresses appetite, slows digestion, and reduces caloric absorption.
Front 2 — Increase Burn: GCG receptor activation raises resting energy expenditure and drives direct fat oxidation in the liver.
Single-agonist compounds only address the first front. Dual agonists add more power to the first front. Retatrutide opens the second front entirely through glucagon receptor activation, a mechanism unavailable to earlier compounds.
Once retatrutide binds to all three receptors, it triggers several overlapping metabolic effects:
Appetite Suppression: GLP-1 pathway activation signals the hypothalamus to reduce hunger drive. Slowed gastric emptying prolongs satiety, reducing overall caloric intake.
Increased Resting Energy Expenditure: Glucagon receptor activation instructs metabolic systems to burn more energy even in the absence of physical activity. Resting metabolic rate is measurably elevated.
Hepatic Fat Reduction: The glucagon receptor mechanism directly drives hepatic lipid oxidation. Research data demonstrates a significant reduction in liver fat content, making retatrutide particularly relevant to fatty liver research.
Lipolysis in Adipose Tissue: Retatrutide surpasses native GIP in stimulating lipolysis in adipocytes. Fat cells break down stored lipids more actively, contributing to greater total fat mass reduction.
Lean Mass Preservation Research data suggest the ratio of fat loss to lean mass loss is comparable to other metabolic interventions. The increased weight loss does not appear to come at the cost of disproportionate lean tissue reduction.
The retatrutide mechanism creates a comprehensive glucose regulation system through its triple receptor approach:
This layered approach to glycaemic control is one reason the compound is studied across both obesity and metabolic disease research.
Retatrutide is formulated for subcutaneous injection. After administration, it enters the bloodstream and binds to GLP-1, GIP, and glucagon receptors throughout the body. Its effects are dose-dependent; higher doses produce stronger receptor responses. Research has tested doses of 1 mg, 4 mg, 8 mg, and 12 mg weekly.
For dose estimation, a retatrutide dosage calculator can help with weekly research calculations.
The half-life of approximately six days means the compound remains biologically active for nearly the entire interval between doses. This consistent receptor activation drives sustained metabolic effects without requiring daily dosing.
The compound undergoes hepatic metabolism and does not interact with the CYP450 enzyme system, lowering the potential for interactions with other compounds.
The retatrutide mechanism supports a range of therapeutic research areas beyond metabolic regulation:
| Compound | Receptors Targeted | Primary Metabolic Action |
| Semaglutide | GLP-1 only | Appetite reduction, gastric slowing |
| Tirzepatide | GLP-1 + GIP | Appetite + insulin regulation |
| Retatrutide | GLP-1 + GIP + GCG | Appetite + insulin + energy expenditure |
The evolution from single to dual to triple hormone agonism is not incremental progress. It represents a fundamentally new biochemical approach to metabolic regulation.
Retatrutide activates three hormone receptors at once: GLP-1, GIP, and glucagon. GLP-1 and GIP suppress appetite and stimulate insulin release. The glucagon receptor raises resting energy expenditure and drives fat oxidation. All three operate simultaneously from a single weekly injection.
Retatrutide is built on incretin pharmacology, the science of gut hormones that regulate insulin and energy balance. It extends beyond prior compounds by adding glucagon receptor activation, which directly raises metabolic rate and liver fat oxidation. The insulin-stimulating GLP-1 and GIP pathways neutralise glucagon’s blood sugar risk. The result is a balanced, three-pathway metabolic system.
Each receptor adds a distinct effect that the others cannot provide. GLP-1 and GIP reduce caloric intake. The glucagon receptor raises energy expenditure independently. This opens a second metabolic front unavailable to single or dual agonist compounds, which is why research data shows 24–29% weight reduction versus ~15% for GLP-1-only agents.
Glucagon receptor activation increases resting energy expenditure and drives the liver to oxidise stored fat directly. It also stimulates lipolysis, the breakdown of fat in adipose tissue. Research data shows up to 82.4% liver fat reduction at the 12 mg dose at 24 weeks. This is the mechanism no currently approved compound can replicate.
GLP-1 agonists reduce appetite and slow digestion. Retatrutide includes this fully and goes further. It adds GIP co-agonism at nearly 9 times the potency of native GIP. It then adds glucagon receptor activation for direct energy expenditure, something no approved GLP-1 compound provides. The difference is mechanistic, not just a matter of dose.
After injection, retatrutide binds to GLP-1, GIP, and glucagon receptors throughout the body. It can lower hunger, delay digestion, and support insulin secretion. At the same time, it raises resting metabolic rate and prompts the liver to burn stored fat. Its six-day half-life sustains all three effects across the entire weekly dosing interval.
Retatrutide is not yet approved by any regulatory authority as of 2026. It is currently in Phase 3 TRIUMPH clinical trials. No approved compound replicates its full three-receptor mechanism. Any product marketed as containing retatrutide before approval is unregulated and should be treated with caution.
The retatrutide mechanism marks a clear advancement in metabolic science. It builds on the biochemical foundation of GLP-1 compounds and dual agonists, then extends further by adding glucagon receptor activation, opening a second metabolic front that earlier compounds could not reach.
The result is a compound that:
The compound’s molecular design, a 39-amino acid helical peptide with a C20 fatty acid modification, enables all three receptor activations from a single molecule, with a six-day half-life supporting once-weekly dosing.
The biochemical evidence supporting the retatrutide mechanism continues to grow. Its triple-receptor approach represents a new frontier in the scientific understanding of metabolic disease and how it may be addressed at the receptor level.
Jastreboff, A. M., et al. (2023). Triple–hormone-receptor agonist retatrutide for obesity a phase 2 trial. New England Journal of Medicine, 389(6), 514–526.
Sanyal, A. J., et al. (2024). Retatrutide for metabolic dysfunction–associated steatotic liver disease. Nature Medicine, 30(8), 2099–2109.
Rosenstock, J., et al. (2023). Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: A phase 2 trial. The Lancet, 402(10401), 529–544.
Coskun, T., et al. (2022). LY3437943, a novel triple GIP, GLP-1, and glucagon receptor agonist: From discovery to clinical proof of concept. Cell Metabolism, 34(9), 1234–1247.

Every peptide vial in a research laboratory is labelled in

The most common peptide mixing mistakes are reported in research

Knowing how to store reconstituted peptides correctly is critical for

Retatrutide is a triple hormone receptor agonist. It works by

At Ignite Peptides, we provide a secure and reliable way to source high-quality research peptides online. As a trusted U.S.-based peptide supplier, we focus on lab-verified products, full transparency, and consistent quality. With fast shipping and responsive support, we’re committed to advancing safe, dependable research you can trust.
This helps us maintain strict standards for regulatory compliance, researcher verification, and responsible product access.