
CagriSema Research: Combining Cagrilintide and Semaglutide Dosing
CagriSema is Novo Nordisk’s investigational fixed-dose combination of cagrilintide 2.4
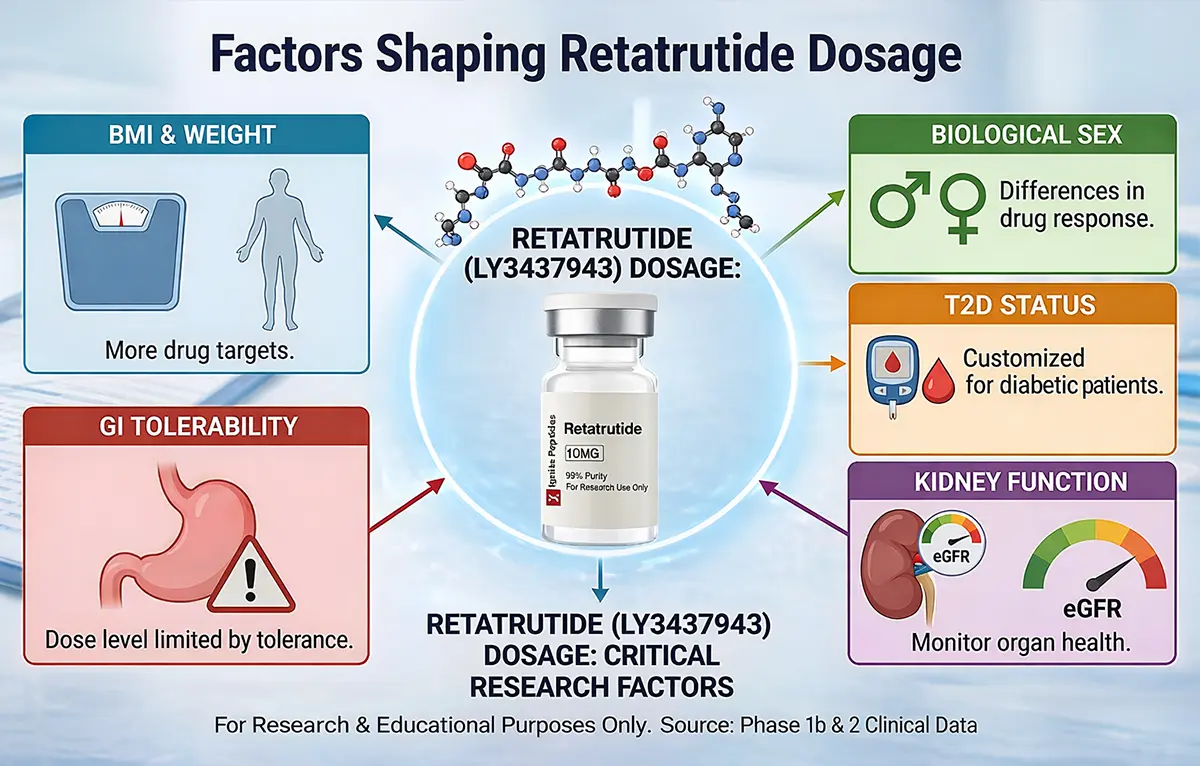
Several factors shape retatrutide dosage in clinical research. These include BMI, biological sex, diabetes status, gastrointestinal (GI) tolerance, kidney function, age, and the trial protocol design itself. No single factor works alone. A full breakdown follows.
Retatrutide (LY3437943) is a triple-receptor hormone agonist created by Eli Lilly. It targets three key receptors: GLP-1 (glucagon-like peptide-1), GIP (glucose-dependent insulinotropic polypeptide), and glucagon. Phase 2 trials showed remarkable results. Participants on the 12 mg dose lost up to 24.2% of their body weight in 48 weeks.
But here is something important to understand. Not everyone on the same dose got the same result. Some lost more. Some tolerated the dose better. Some needed the dose adjusted. Why does this happen?
The answer lies in the retatrutide dose factors that researchers and clinicians track carefully. These are the biological, medical, and pharmacokinetic (drug behaviour) variables that influence how much retatrutide is used and how well it works in a research setting.
If you want a broader explanation of how dosing works in studies, see the retatrutide dosing research guide.
This article explains each factor clearly. All information comes from published Phase 1b, Phase 2, and meta-analysis data. This is for research and educational purposes only.
BMI is one of the most studied retatrutide dose factors. In the Phase 2 obesity trial published in the New England Journal of Medicine, researchers split participants by BMI. One group had a BMI below 36. The other had a BMI of 36 or higher.
The results were clear. Participants with a higher BMI responded better to the same dose. At the 8 mg dose, the high-BMI group lost 26.5% of body weight. The lower-BMI group lost 21.3%. A similar pattern appeared at 12 mg.
Researchers believe that more adipose (fat) tissue may create a larger pharmacological target. In simple terms, the drug has more to act on. This may amplify the metabolic response without requiring a higher dose.
It is important to note that BMI does not determine dose in current trials. Researchers use it as a stratification variable, a way to group and compare participants fairly. However, this data may shape future dosing guidelines once Phase 3 results arrive.
| Dose Group | BMI < 35 Weight Loss | BMI ≥ 35 Weight Loss |
| 8 mg/week | 21.3% | 26.5% |
| 12 mg/week | 21.5% | 26.4% |
Source: NEJM Phase 2 Obesity Trial, 48-week data
Researchers often summarise this type of data using structured reference tables.
A detailed retatrutide dosage chart for research studies explains how dosing tiers are typically structured across trials.
Biological sex is another key retatrutide dose factor. The Phase 2 trial enrolled roughly equal numbers of men and women; about 52% were male. This was done on purpose. Researchers wanted to test for sex-based differences in dose response.
The findings were striking. At the 8 mg dose, women lost an average of 28.5% of their body weight. Men lost 19.8%. At 12 mg, women lost 26.6%, and men lost 21.9%.
The exact reason is still under investigation. Researchers suggest three possible explanations:
This sex-based difference in weight loss is not unique to retatrutide. Post-hoc analyses of other GLP-1 receptor agonists have shown similar patterns. The NEJM authors noted this and designed the retatrutide trial to account for it.
At this stage, biological sex does not change the research dose used. Both sexes follow the same titration schedule. But these findings may influence future personalised medicine dosing decisions.
Diabetes status is one of the clearest retatrutide dose factors. Researchers ran two separate Phase 2 trials, one for people with obesity but no diabetes, and one for people with type 2 diabetes. The protocols were different. The outcomes were different, too.
The T2D trial, published in The Lancet Diabetes and Endocrinology, enrolled 281 participants. Their HbA1c (a measure of blood sugar control) ranged from 7.0% to 10.5%. The study ran for 36 weeks, shorter than the 48-week obesity trial.
One key difference was the starting dose. The T2D trial included a 0.5 mg fixed dose group. This very low starting point was not used in the obesity trial. It reflects extra caution around blood glucose management in diabetic participants.
Weight loss outcomes were also lower. At 12 mg, T2D participants lost an average of 16.94% of body weight at 36 weeks. Non-diabetic participants lost around 24.2% at 48 weeks. This difference is consistent with similar patterns seen with other incretin therapies.
| Comparison Point | Non-Diabetic Trial | T2D Trial |
| Trial Duration | 48 weeks | 36 weeks |
| Lowest Dose Tested | 1 mg (fixed) | 0.5 mg (fixed) |
| Max Weight Loss at 12 mg | ~24.2% | ~16.94% |
| HbA1c Reduction | Not the primary endpoint | 1.3–2.0% reduction |
Source: NEJM Phase 2 (non-diabetic) | The Lancet Phase 2 (T2D)
Glycaemic (blood sugar) benefit was strong in the T2D group. HbA1c fell by 1.3% to 2.0% with 4–12 mg doses. This outperformed the active comparator dulaglutide 1.5 mg. The key takeaway: diabetes status changes both the dosing protocol and the expected outcomes in research.
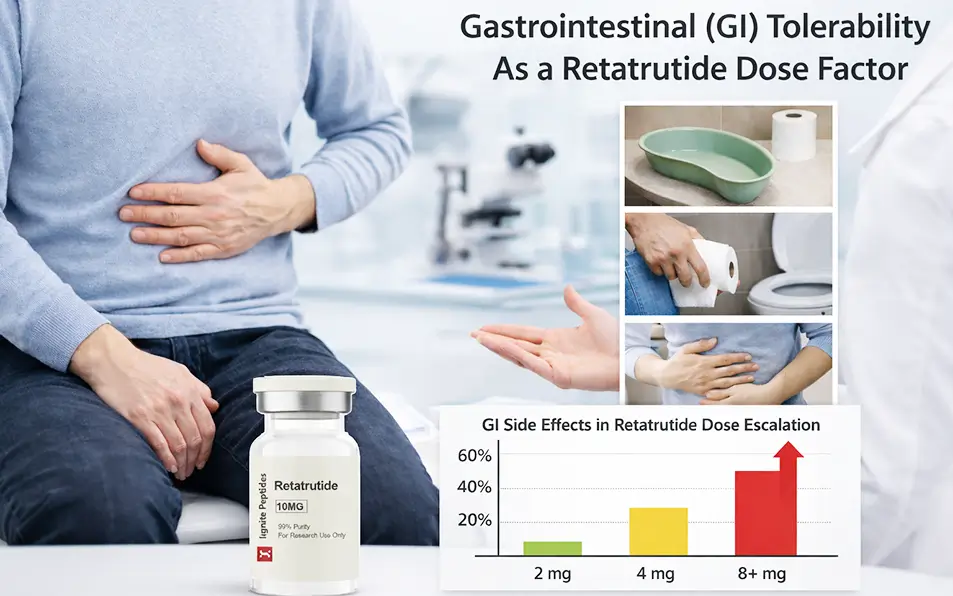
This is arguably the most practical of all retatrutide dose factors. GI side effects, such as nausea, diarrhoea, vomiting, and constipation, were the most common reason for dose adjustment in all retatrutide trials.
Retatrutide slows gastric emptying. This mechanism helps explain its effect on appetite control. But when the dose rises too fast, the GI system cannot adapt quickly enough. The result is discomfort that can force a dose hold or reduction.
In the Phase 2 obesity trial, GI adverse events occurred in 50–60% of participants at higher doses. These were mostly mild to moderate. Serious GI events were rare.
The starting dose made a significant difference. Participants who began at 2 mg had fewer GI events than those who started at 4 mg. Crucially, this did not hurt their long-term weight loss outcomes.
Key Research Finding: Starting at 2 mg per week rather than 4 mg significantly reduced GI side effects. Long-term weight loss outcomes remained the same. This finding directly influenced the dose escalation design for Phase 3 TRIUMPH trials.
The standard titration schedule used in Phase 2 studies moved doses up every four weeks. For example: 2 mg for weeks 1–4, then 4 mg, then 8 mg, then 12 mg. This four-week interval was found to be the tolerability sweet spot.
Researchers who want to understand how escalation stages are structured can review this retatrutide dosage schedule guide.
Participants who could not tolerate escalation stayed at their current dose longer. This means the final dose reached is itself a product of individual GI tolerance, not just a fixed protocol decision.
Kidney function is a critical but often overlooked retatrutide dose factor. Both retatrutide trials required participants to have an estimated glomerular filtration rate (eGFR) of at least 45 ml/min/1.73 m². People with more severe kidney impairment were excluded.
This means current dosing data does not cover patients with moderate-to-severe chronic kidney disease (CKD). Future approved labelling is expected to include specific guidance for this group.
A 2025 post-hoc analysis published in Kidney International Reports assessed kidney parameters across both trials. The findings were dose-dependent.
Safety monitoring during trials tracked liver enzymes (ALT, AST) and kidney markers throughout. Any significant abnormality could trigger a dose hold. This means kidney health acts as an ongoing gate on achievable dose, not just a baseline eligibility criterion.
The Phase 2 trials enrolled adults aged 18 to 75 years. No older adult-specific subgroup analysis has been published yet. However, age is still a relevant retatrutide dose factor, especially when extrapolating to real-world clinical use.
As people age, kidney and liver function naturally decline. This affects how the body processes any drug. In older adults, the pharmacokinetic (drug behaviour) environment is different from that of a 35-year-old.
Retatrutide is metabolised primarily in the liver. It does not interact with cytochrome P450 enzymes, which reduces the risk of drug-drug interactions. This is an advantage. But GI effects in older adults carry a different risk profile.
Nausea and diarrhoea can cause dehydration. In younger people, this is manageable. In older adults, dehydration increases the risk of falls, dizziness, and electrolyte imbalances.
For older adults, the anticipated approach based on similar incretin drugs is: start lower, increase more slowly. This is sometimes called the “start low, go slow” principle. It prioritises tolerability over rapid dose escalation.
Note for Researchers: Older participants may need extended time at each dose level before moving up. Current trial protocols allow for this. Participants can remain at a dose tier longer if escalation is not yet tolerated.
Understanding retatrutide’s pharmacokinetics (PK) is essential to understanding its dosing. The PK profile directly shapes how doses are structured in research studies.
For a broader overview of the compound’s mechanisms and study background, see the complete retatrutide research guide.
Retatrutide has dose-proportional pharmacokinetics. This means plasma drug concentration rises predictably as the dose increases. There is no unexpected accumulation at higher doses. This makes dose escalation planning more reliable.
| PK Property | Research Significance |
| Half-life: ~6 days | Supports once-weekly subcutaneous injection; steady-state reached in 4–5 weeks |
| Tmax: 12–72 hours post-dose | Peak concentration after injection; explains injection-day GI effects in some participants |
| Dose-proportional | Plasma levels scale linearly with dose, no unexpected accumulation, easier safety modelling |
| Hepatic metabolism | No cytochrome P450 interactions, lower drug-drug interaction risk |
| No renal excretion | Reduces kidney-specific dosing concerns, though renal eligibility threshold still applies |
Source: Phase 1 PK study (Singapore cohort); NEJM Phase 2 supplementary data
The six-day half-life is particularly important. It means that missing one weekly dose does not immediately drop plasma levels to zero. However, consistency matters for steady-state concentration. Irregular dosing can affect both efficacy and tolerability patterns.
This factor is unique to research settings. In a clinical trial, participants do not simply receive whatever dose seems best for them individually. They are randomised to a pre-set dose group. The protocol itself is therefore one of the most direct retatrutide dose factors in a research context.
The Phase 2 obesity trial used seven arms. Participants were randomly assigned to receive: placebo, 1 mg (fixed), 4 mg (with escalation from 2 mg), 4 mg (without escalation), 8 mg (with escalation from 2 mg), 8 mg (with escalation from 4 mg), or 12 mg (with escalation from 2 mg).
The inclusion of both escalation and non-escalation arms was deliberate. It lets researchers directly compare how the starting dose speed affects tolerability. The result was clear evidence that starting at 2 mg, not 4 mg, produced better tolerability without sacrificing long-term weight loss.
This research finding will likely define the future approved dosing label. So the protocol design does not just describe how doses were used; it generated the evidence that shapes how doses will be used in clinical practice.
The table below summarises each factor, its clinical significance, and the level of research evidence supporting it.
| Dose Factor | Why It Matters | Evidence Source |
| BMI / Starting Weight | Higher BMI linked to greater % weight loss at the same dose | NEJM Phase 2 sub-analysis |
| Biological Sex | Women showed greater weight loss than men at equal doses | NEJM Phase 2 pre-specified analysis |
| T2D Status | Separate protocol; lower weight loss; strong glycaemic benefit | Lancet Phase 2 T2D trial |
| GI Tolerability | The top cause of dose modification or hold is managed by slow titration | Phase 1b + Phase 2 safety data |
| Kidney Function | eGFR threshold required; monitored throughout; affects dose safety ceiling | Kidney Int. Reports 2025 post-hoc |
| Age / Polypharmacy | Older adults need slower escalation; GI risk compounds dehydration risk | Extrapolation; limited direct data |
| Pharmacokinetics | Dose-proportional; 6-day half-life enables weekly dosing | Phase 1 PK study, Singapore cohort |
| Trial Protocol Design | In research, protocol determines dose, not individual clinical judgement | NEJM + Lancet Phase 2 design |
The highest dose studied is 12 mg per week. The compound is typically delivered as a weekly subcutaneous injection. In the Phase 2 obesity trial, 12 mg produced up to 24.2% mean body weight loss at 48 weeks in non-diabetic participants.
No. BMI is used as a stratification variable, a tool for grouping participants fairly. It does not assign a dose. However, participants with BMI ≥ 35 showed greater weight-loss responses at the same doses as those with lower BMI.
Retatrutide slows gastric emptying. If the dose rises too quickly, the GI system is overwhelmed. This causes nausea and diarrhoea. The four-week escalation interval was validated in Phase 2 as the safest and most effective approach.
Yes. The T2D trial used different dose arms, including a 0.5 mg fixed group not tested in the obesity trial. T2D participants also had lower weight-loss outcomes at equivalent doses, though glycaemic control improved significantly.
Yes. Trials required an eGFR of at least 45 ml/min/1.73 m². People with worse kidney function were excluded. At higher doses, retatrutide improved eGFR in obesity participants and reduced albumin-creatinine ratio in T2D participants.
No. Retatrutide is still an investigational drug. Phase 3 TRIUMPH trials are ongoing. Regulatory approval from the FDA is not anticipated before 2027. All dosing in this article reflects research protocols only.
Retatrutide dose factors do not operate in isolation. A high-BMI woman with no diabetes and good GI tolerance will have a very different research dose journey than an older man with early-stage kidney disease and type 2 diabetes. Both may be on the same trial. But their dose trajectory and their results will likely look different.
This is exactly why dose-dependent weight loss pharmacotherapy research is so important. It involves more than simply choosing a number. It is about understanding which biological and clinical variables shape the dose-response relationship for each type of participant.
The TRIUMPH Phase 3 trials are now underway. They will generate more sub-group data across larger and more diverse populations. That data will be the foundation for personalised medicine dosing guidelines once retatrutide receives regulatory approval.
Researchers sometimes use digital tools to model dose escalation scenarios and estimate study protocols.
For example, the retatrutide dosage calculator helps illustrate how weekly dosing schedules may be structured in research settings.
Until then, retatrutide remains an investigational compound. All dosing decisions remain within the strict boundaries of structured research protocols, not individual clinical judgement.
What to Read Next→ How Does Retatrutide Work? (GLP-1/GIP/Glucagon Receptor Mechanism)→ Retatrutide Phase 3 TRIUMPH Trials: What to Expect→ Retatrutide vs Tirzepatide: Triple vs Dual Agonist Compared→ Managing Retatrutide Side Effects in Research Participants
Disclaimer: Retatrutide (LY3437943) is an investigational compound. It is not FDA-approved. All dosing information in this article reflects clinical research protocols only, not prescribing guidance. Seek advice from a licensed healthcare professional before making medical decisions.

CagriSema is Novo Nordisk’s investigational fixed-dose combination of cagrilintide 2.4

Semaglutide is one of the most studied GLP-1 receptor agonists

Introduction Designing a precise GLP-1 titration schedule is one of
Introduction Research on the GLP-1 mechanism is one of the

At Ignite Peptides, we provide a secure and reliable way to source high-quality research peptides online. As a trusted U.S.-based peptide supplier, we focus on lab-verified products, full transparency, and consistent quality. With fast shipping and responsive support, we’re committed to advancing safe, dependable research you can trust.
This helps us maintain strict standards for regulatory compliance, researcher verification, and responsible product access.