
Common Peptide Mixing Mistakes and How to Avoid Them
The most common peptide mixing mistakes are reported in research
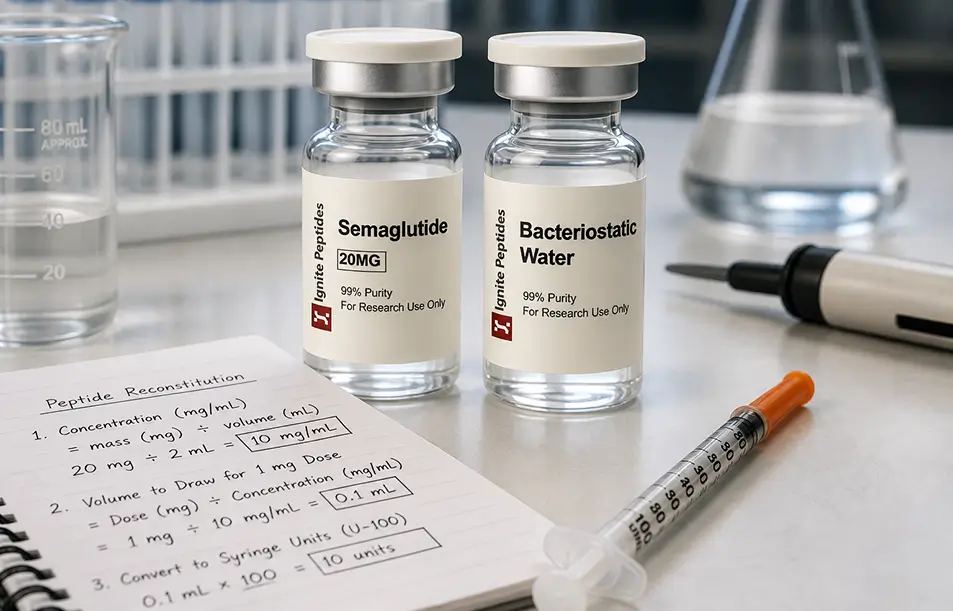
Every peptide vial in a research laboratory is labelled in milligrams (mg). But the moment you add a diluent, you work in millilitres (mL). These two measurements represent different properties. One is mass. The other is volume. You cannot swap them without a formula.
Knowing how to calculate mg to mL is a core skill in peptide reconstitution. Many researchers use a peptide reconstitution calculator to verify their numbers before drawing from a vial. Get it right, and your solution concentration is accurate. Get it wrong, and every aliquot drawn from that vial carries the error forward through your entire study.
This step-by-step guide covers the core formula, worked examples, a unit conversion table, diluent selection, and storage guidelines. All content is written for in vitro laboratory research use only.
Quick Answer: Volume (mL) = Desired Dose (mg) ÷ Concentration (mg/mL). To find concentration: Concentration (mg/mL) = Peptide Mass (mg) ÷ Diluent Volume (mL). Both values are set when you reconstitute your peptide vial.
This is the first thing every researcher must understand. mg and mL are not equal units, and they are not interchangeable.
A milligram (mg) measures mass, or how much substance is present.
Millilitres (mL), on the other hand, measure volume, which refers to the space a liquid occupies.
To move between them, you need the density of the solution. Density links mass and volume together.
The formula is: mL = mg ÷ (density × 1,000). For pure water, density = 1 g/mL. So 1 mL of water equals 1,000 mg, not 1 mg.
For aqueous peptide solutions reconstituted with bacteriostatic water (BAC water), density is approximately 1 g/mL. This makes the maths simpler. You can treat the solution as having water-equivalent density in most research settings.
Key Fact: 1 mL of water ≈ 1,000 mg. For peptide solutions in BAC water, the density ≈ is 1 g/mL. There is no fixed 1:1 ratio between mg and mL.
There are two formulas you need. Use them in sequence.
Concentration (mg/mL) = Peptide Mass (mg) ÷ Diluent Volume (mL)
This formula is applied once, at the moment you add your diluent to the lyophilised powder. The result is your working solution concentration.
Example: A 10 mg vial + 2 mL BAC water = 5 mg/mL concentration.
To find volume, use this formula: divide the required dose (in mg) by the concentration (in mg per mL).
This formula is applied each time you prepare an aliquot. It tells you exactly how many mL to draw for your target dose.
Example: A target dose of 1 mg from a 5 mg/mL solution = 1 ÷ 5 = 0.2 mL.
Important unit note:
Peptide vials are labelled in mg. But research protocols often express doses in micrograms (mcg). Make sure all values are in matching units before performing any calculation.
1 mg = 1,000 mcg. To convert mcg to mg, divide by 1,000. To convert mg to mcg, multiply by 1,000.
Unit Conversion Quick Reference
| From | To | Operation | Example |
|---|---|---|---|
| mg | mcg | × 1,000 | 2 mg → 2,000 mcg |
| mcg | mg | ÷ 1,000 | 500 mcg → 0.5 mg |
| mg/mL | mcg/mL | × 1,000 | 5 mg/mL → 5,000 mcg/mL |
| mL | U-100 syringe units | × 100 | 0.2 mL → 20 units |
| U-100 units | mL | ÷ 100 | 25 units → 0.25 mL |
Below is a full worked example. Follow each step in order. All values are for research purposes only.
Scenario: A researcher has a 5 mg lyophilised peptide vial. They add 2 mL of bacteriostatic water. Their research protocol targets a dose of 0.5 mg per aliquot.
Result: Draw to the 20-unit mark on a U-100 syringe to deliver 0.5 mg from a 2.5 mg/mL solution.
Concentration: 10 mg ÷ 1 mL = 10 mg/mL.
Target dose: 1 mg. Volume to draw: 1 mg ÷ 10 mg/mL = 0.1 mL = 10 units on a U-100 syringe.
Notice: the same 10 mg vial with 2 mL of water gives 5 mg/mL. The peptide mass does not change. Only the concentration changes. This is the Law of Conservation of Mass at work in the lab.
If you are working with a specific peptide such as Retatrutide, refer to the Retatrutide dosage calculation guide which applies the same mg to mL formula to that compound specifically.
The diluent you choose affects both concentration accuracy and solution stability. The most widely used option in peptide research is bacteriostatic water.
BAC water includes 0.9% benzyl alcohol to prevent bacterial growth. This preservative prevents bacterial growth. It allows multi-dose use of the same vial for up to 28 days. BAC water is the standard choice for most in vitro peptide research.
Sterile water is highly pure with no preservatives. It must be used within 24 to 48 hours after opening. It is better suited for cell culture assays where benzyl alcohol may affect cell viability.
Dilute acetic acid (0.1–1%) is used for peptides with poor aqueous solubility. Some hydrophobic sequences do not dissolve well in pure water. A dilute acid environment improves solubility for these compounds.
| Diluent | Best Use Case | Stability Window | Key Note |
|---|---|---|---|
| Bacteriostatic Water (BAC) | General peptide research, multi-dose | Up to 28 days at 2–8°C | Gold standard for most peptides |
| Sterile Water | Cell culture, cytotoxicity studies | 24–48 hours | Single-use only, no preservative |
| 0.1% Acetic Acid | Poorly soluble or hydrophobic peptides | Varies by peptide class | Check the peptide solubility profile first |
| DMSO | Highly hydrophobic compounds | Varies | Dilute with aqueous buffer before use |
Not sure how much BAC water to add for your target concentration? Use the bacteriostatic water calculator to get the exact volume in seconds.
Research Note: Bacteriostatic water and sterile water both have a density ≈ of 1 g/mL. This means your mg to mL formula applies equally to both diluents in standard peptide reconstitution.
Accurate concentration calculation means nothing if the peptide degrades before use. Storage conditions directly impact solution stability and research reproducibility.
Lyophilised (dry) peptides: Store at −20°C for long-term stability. Short-term storage (under 2 weeks) is acceptable at 4°C. Always allow sealed vials to reach room temperature before opening. This prevents condensation from entering the vial.
Reconstituted solutions: Refrigerate at 2–8°C. Solutions prepared with BAC water remain stable for up to 30 days. Solutions in sterile water must be used within 24 to 48 hours.
Freeze-thaw cycles: Repeated freezing and thawing break down peptide structural integrity. If long-term storage is needed, divide the reconstituted solution into small aliquots before freezing. Use each aliquot once and discard.
For a complete storage protocol covering temperature, light exposure, and labelling practices, read the full guide on how to store peptides after reconstitution.
Light exposure: Some peptide sequences are photosensitive. Store in amber vials or wrap vials in aluminium foil to prevent light-induced oxidation.
| Peptide State | Temperature | Duration | Notes |
|---|---|---|---|
| Lyophilised powder (sealed vial) | −20°C | Up to 24 months | Allow to reach room temp before opening |
| Lyophilised powder (short-term) | 4°C | Up to 2 weeks | Keep dry and sealed |
| Reconstituted in BAC water | 2–8°C | Up to 30 days | Label with date and concentration |
| Reconstituted in sterile water | 2–8°C | 24–48 hours | Single-use protocol only |
| Aliquots (frozen) | −20°C | 3–4 months | No repeated freeze-thaw cycles |
Small calculation errors compound quickly in laboratory work. A 10% volume error at reconstitution propagates as a 10% dosing error across every aliquot in a study. These overlap with broader common peptide mixing mistakes that researchers encounter during preparation. Below are the most common mistakes and how to avoid them.
First, calculate concentration: Concentration (mg/mL) = Peptide Mass (mg) ÷ Diluent Volume (mL). Then calculate the draw volume: Volume (mL) = Desired Dose (mg) ÷ Concentration (mg/mL). Both steps are required. You cannot skip the concentration calculation.
No. mg is mass. mL is volume. For water, 1 mL = 1,000 mg (because water density = 1 g/mL). For a peptide solution, 1 mL contains the solvent mass plus the dissolved peptide mass. There is no direct 1:1 relationship.
Approximately 1,000 mg of total solution per mL (density ≈ 1 g/mL). The dissolved peptide contributes only a small fraction of that mass. For a 5 mg/mL peptide solution, 1 mL contains 5 mg of peptide and approximately 995 mg of water-based solvent.
There is no universal standard. Most researchers target a concentration that produces convenient draw volumes for their syringe size. Common practice is to add 1 to 3 mL of BAC water per vial. Use the concentration formula to calculate the resulting mg/mL, or run your numbers through the peptide calculator to verify before reconstituting.
The total peptide mass in the vial stays the same. Adding more water lowers the concentration. This means each dose requires a larger draw volume. It does not damage the peptide. Recalculate your concentration with the actual volume added and proceed.
Yes. Convert mcg to mg first by dividing by 1,000. Then apply the standard formula: Volume (mL) = Dose (mg) ÷ Concentration (mg/mL). The formula works with any consistent unit pair as long as the dose and concentration use t
| Step | Action | Formula |
|---|---|---|
| 1 | Identify vial content | Read label (mg) |
| 2 | Add measured diluent volume | Record exact mL added |
| 3 | Calculate concentration | Concentration = mg ÷ mL |
| 4 | Identify the target dose | From research protocol (mg or mcg) |
| 5 | Calculate the draw volume | Volume = Dose ÷ Concentration |
| 6 | Convert to syringe units (if U-100) | Units = mL × 100 |
Knowing how to calculate mg to mL is a foundational research skill. It is not complicated. But it must be done correctly every time.
The two formulas are straightforward. Concentration = mass divided by volume. Draw volume = dose divided by concentration. Apply them in sequence. Use consistent units throughout. Verify your diluent volume before reconstituting.
Accurate reconstitution protects the integrity of your research data. Decimal errors and unit confusion are the most common causes of bad results in peptide studies. A small check at every step prevents large errors downstream.
Label every vial with the compound name, reconstitution date, and final concentration. Store solutions according to the guidelines in this article. Use aliquots to avoid freeze-thaw degradation.
Research Disclaimer: All content in this article is intended for in vitro and laboratory research use only. This information does not constitute medical advice, clinical guidance, or instructions for human or veterinary use. For research purposes only.
The following peer-reviewed sources support the scientific content in this article. All sources were retrieved from PubMed.
1. Izutsu K-I. (2018). Applications of Freezing and Freeze-Drying in Pharmaceutical Formulations. Advances in Experimental Medicine and Biology, 1081, 371-383. This review establishes that lyophilisation significantly improves the storage stability of peptides by removing water and reducing degradation pathways. DOI: 10.1007/978-981-13-1244-1_20
2. O’Fagain C & Colliton, K. (2023). Storage and Lyophilisation of Pure Proteins. Methods in Molecular Biology, 2699, 421-475. This chapter outlines best practices for lyophiliser use, freezing stages, and protein stability maintenance during long-term storage. DOI: 10.1007/978-1-0716-3362-5_19
3. Kenley RA et al. (2000). Kinetics of Pramlintide Degradation in Aqueous Solution as a Function of Temperature and pH. AAPS PharmSciTech, 1(2), E7. A 30-month stability study showing aqueous peptide solutions degrade based on pH and temperature. Peptides at pH 4.0 showed under 2% purity loss at 5 degrees C, supporting refrigerated storage protocols. DOI: 10.1208/pt010207
4. Arii K, Kai T & Kokuba Y. (1999). Degradation Kinetics of L-Alanyl-L-Glutamine and Its Derivatives in Aqueous Solution. European Journal of Pharmaceutical Sciences, 7(2), 107-112. Characterises hydrolysis and deamination as primary degradation pathways in aqueous peptide solutions, supporting the need for accurate concentration control at reconstitution. DOI: 10.1016/s0928-0987(98)00012-8

The most common peptide mixing mistakes are reported in research

Knowing how to store reconstituted peptides correctly is critical for

Retatrutide is a triple hormone receptor agonist. It works by

For decades, metabolic pharmacology focused on one goal: reducing calorie

At Ignite Peptides, we provide a secure and reliable way to source high-quality research peptides online. As a trusted U.S.-based peptide supplier, we focus on lab-verified products, full transparency, and consistent quality. With fast shipping and responsive support, we’re committed to advancing safe, dependable research you can trust.
This helps us maintain strict standards for regulatory compliance, researcher verification, and responsible product access.