
GLP-1 Titration Schedule Week-by-Week Research Dosing Protocol for GLP-1(S)
Introduction Designing a precise GLP-1 titration schedule is one of

Research Disclaimer: This article is for informational and research purposes only. All products sold by Ignite Peptides are chemical reagents for licensed research use only. They are not intended for human consumption, diagnosis, or medical treatment.
In December 2025, Eli Lilly announced something the research world had not seen before.
Participants in the TRIUMPH-4 Phase 3 trial lost an average of 28.7% of their body weight at 68 weeks. That is roughly 71 pounds per person. No obesity pharmacotherapy had ever produced that number in a Phase 3 trial.
The compound responsible for? Retatrutide is a new compound that targets three different hormone receptors.
Semaglutide, meanwhile, remains the gold standard. It is FDA-approved, widely prescribed, and backed by years of real-world safety data. For most researchers and clinicians, it is still the benchmark.
So where does that leave the retatrutide vs semaglutide comparison in 2026?
This retatrutide vs semaglutide guide breaks it all down. We cover mechanisms, clinical data, side effects, FDA status, and what each compound offers for metabolic research. The comparison is based on the latest published trial data, including the December 2025 TRIUMPH-4 readout.
Here’s a fast look at the key differences:
| Feature | Retatrutide | Semaglutide |
| Mechanism | Triple agonist: GLP-1, GIP, glucagon | Single agonist: GLP-1 only |
| Best Phase 3 weight loss | 28.7% at 68 weeks (TRIUMPH-4) | 14.9% at 68 weeks (STEP 1) |
| FDA status | Phase 3 trials ongoing | Fully approved (Wegovy, Ozempic) |
| Results onset | 8–12 weeks | 12–16 weeks |
| Unique side effect | Dysesthesia (up to 20.9% at 12 mg) | Gallbladder issues (rare) |
| Cardiovascular data | Markers improved; outcomes trial pending | SELECT trial: 20% MACE reduction |
| Approval timeline | NDA filing expected late 2026 | Approved since 2021 (obesity) |
| Research availability | Available for licensed research use | Available for licensed research use |
Key takeaways:
Retatrutide (scientific name: LY3437943) is an investigational peptide developed by Eli Lilly. It is a once-weekly subcutaneous injection. What makes it different from every other GLP-1 compound is its triple mechanism.
It activates three separate hormone receptors in a single molecule:
Semaglutide only targets the first pathway. Retatrutide targets all three simultaneously. That is why the weight loss numbers are so different.
Because retatrutide comes as a compounded vial requiring reconstitution, preparing each dose involves unit conversions that semaglutide pen users never need to perform. The retatrutide dosing calculator guide explains this process in full.
The TRIUMPH program is Eli Lilly and Company’s Phase 3 study plan for retatrutide. It includes multiple trials across obesity, type 2 diabetes, knee osteoarthritis, sleep apnea, chronic lower back pain, and liver disease.
The first Phase 3 readout, TRIUMPH-4, was published on December 11, 2025.
Here is what the data showed:
Additional Phase 3 trial results are expected to be released during 2026. If results hold, Lilly is expected to file for FDA approval in late 2026. Analysts project an approval window of early to mid-2027.
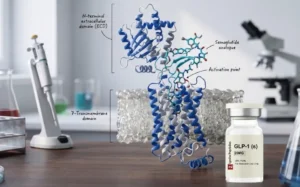
Semaglutide is a synthetic analogue of GLP-1, a gut hormone released after eating. It has a half-life of roughly one week, which is why it is dosed once weekly.
When it binds to GLP-1 receptors, it does three things:
These effects combine to reduce caloric intake and improve blood sugar regulation. The mechanism is well understood and has been studied in thousands of patients across multiple Phase 3 and 4 trials.
The FDA approved Semaglutide under two brand names:
It has the deepest real-world safety dataset of any GLP-1 compound. Millions of patients have used it. Its side effects are well studied and understood.
The SELECT trial (2023) added another layer of credibility. It was the first obesity trial to show that a weight-loss drug could reduce major cardiovascular events, specifically, a 20% reduction in MACE in non-diabetic adults with obesity and existing cardiovascular disease. That result changed how the medical community thinks about obesity pharmacotherapy.
Retatrutide does not yet have equivalent cardiovascular outcomes data. That gap matters for research contexts focused on cardiometabolic endpoints.

Both peptides affect appetite and blood sugar. But their mechanisms diverge significantly beyond that.
Semaglutide works on the input side of the energy equation. It reduces hunger and slows digestion. This effect may help reduce overall food intake.
Retatrutide works on both sides at once. The GLP-1 and GIP components reduce appetite and intake. But the glucagon receptor component drives the body to expend more energy through thermogenesis and hepatic fat metabolism. It is attacking the energy balance from two directions.
The glucagon receptor component is what separates retatrutide from every other compound in this class.
Glucagon normally rises during fasting to stimulate the liver to release stored glucose. But in retatrutide, glucagon receptor activation is balanced against concurrent GLP-1 activity, which controls blood sugar, so glucose levels stay stable. Meanwhile, the thermogenic and fat-burning effects of glucagon are preserved.
This mechanism also explains why retatrutide shows promise in metabolic dysfunction-associated steatotic liver disease (MASLD). Hepatic fat clearance is a direct result of glucagon receptor activation. Eli Lilly has an ongoing TRIUMPH trial specifically studying this indication.
The retatrutide story has two chapters. The Phase 2 results (published in NEJM, 2023) showed an average weight loss of 24.2% at 48 weeks. Those numbers were already unprecedented for a compound in development.
The Phase 3 TRIUMPH-4 results go further. At 68 weeks, the 12 mg dose produced 28.7% average weight loss. That is the highest recorded in any Phase 3 obesity pharmacotherapy trial to date.
For comparison:
| Compound | Mechanism | Weight Loss | Study Duration |
| Semaglutide | GLP-1 | ~14.9% | 68 weeks (STEP 1) |
| Tirzepatide | GLP-1 + GIP | ~20.9% | 72 weeks (SURMOUNT-1) |
| Retatrutide | GLP-1 + GIP + GcgR | ~28.7% | 68 weeks (TRIUMPH-4) |
The progression is striking. Each additional receptor target appears to meaningfully increase the weight loss outcome.
Both compounds improve markers beyond body weight.
Semaglutide has the most comprehensive dataset here. It reduces HbA1c, LDL cholesterol, blood pressure, and critically, cardiovascular event rates. The SELECT trial remains the defining evidence that a weight-loss compound can reduce heart attack and stroke risk.
Retatrutide showed strong cardiometabolic improvements in TRIUMPH-4. Triglycerides, non-HDL cholesterol, hsCRP, and systolic blood pressure all decreased. The systolic blood pressure reduction at the 12 mg dose was 14.0 mmHg, clinically significant. Full cardiovascular outcomes trial data are expected through the TRIUMPH program in 2026.
One important caveat for any retatrutide vs semaglutide comparison: these trials were not head-to-head. Different study populations, different designs. Direct comparison of the numbers has limits. Researchers should factor this into any protocol design that uses these studies as reference points.
The side-effect profiles of both compounds overlap significantly. The GI symptoms are shared across the entire incretin class.
Common side effects for both:
| Side Effect | Retatrutide (12 mg) | Semaglutide |
| Nausea | 43.2% | ~44% (STEP 1) |
| Diarrhea | 33.1% | ~30% |
| Constipation | 25.0% | ~24% |
| Vomiting | 20.9% | ~24% |
| Decreased appetite | 18.2% | ~19% |
Both compounds require gradual dose escalation to manage these effects.
This is the detail that sets TRIUMPH-4 apart from the Phase 2 data, and from semaglutide entirely.
Dysesthesia is an abnormal sensation on the skin. It can feel like tingling, burning, numbness, or unusual sensitivity to touch. It is not a dangerous condition. But it was not observed at meaningful levels in retatrutide’s Phase 2 trial. It emerged in Phase 3.
In TRIUMPH-4, dysesthesia occurred in 8.8% of participants on the 9 mg dose and 20.9% on the 12 mg dose, compared with just 0.7% on placebo. Most cases were mild and rarely caused participants to stop treatment.
Why did it appear in Phase 3 and not Phase 2? The honest answer is that researchers do not yet know. It may be related to the higher doses studied (9 mg and 12 mg vs. lower Phase 2 doses) or to the longer trial duration. Seven more readouts are coming in 2026. Analysts are watching to see if dysesthesia appears across different populations and indications.
For researchers designing protocols, this is an important variable to track and document.
Semaglutide-specific concerns: Gallbladder disease (cholelithiasis) is the most documented serious risk specific to semaglutide. Rare cases of pancreatitis have also been reported across the GLP-1 class.
Semaglutide is fully available today. Physicians can prescribe Wegovy for obesity and Ozempic for type 2 diabetes. It has years of post-approval surveillance data and is one of the most studied weight-loss drugs in history.
Retatrutide is not FDA-approved. It is an investigational compound currently in Phase 3 trials.
Seven additional Phase 3 trials evaluating retatrutide in obesity and type 2 diabetes are expected to be completed in 2026. Based on TRIUMPH-4’s positive results, Lilly is expected to submit a New Drug Application (NDA) to the FDA in late 2026. A realistic approval window is early to mid-2027, though this is subject to how the remaining TRIUMPH readouts land.
These compounds are available for licensed research use only. They are not for human use or clinical administration.
| Semaglutide | Tirzepatide | Retatrutide | |
| Receptors | GLP-1R | GLP-1R + GIPR | GLP-1R + GIPR + GcgR |
| Best Phase 3 weight loss | ~14.9% | ~20.9% | ~28.7% |
| FDA approved | Yes | Yes | No (Phase 3) |
| Cardiovascular outcomes | Yes (SELECT) | Pending | Pending |
| Unique advantage | Most safety data | Dual mechanism | Thermogenesis + energy expenditure |
The pattern here is clear. Each generation adds a receptor. Each addition brings a larger weight-loss effect. Retatrutide is the most potent of the three by efficacy data, but also the least mature by safety evidence.
Tirzepatide sits between the two. It is FDA-approved, has a strong trial record, and offers better efficacy than semaglutide. For researchers who need an approved compound with dual-receptor activity, it is a relevant reference point.
The answer depends entirely on what the research question is.
Semaglutide is the safer bet when reproducibility and regulatory alignment matter most.
Retatrutide offers the most mechanistic novelty in this class right now. For researchers studying the cutting edge of metabolic science, it is the most interesting compound on the market.
For reconstitution support, use the Ignite Peptides Peptide Calculator for accurate volume and concentration calculations, and the Bacteriostatic Water Calculator for reconstitution water volumes.
The retatrutide vs semaglutide comparison in 2026 is not a close call on efficacy. Retatrutide is producing weight loss numbers that no compound in this class has achieved before, 28.7% at 68 weeks in Phase 3. That is a meaningful step beyond semaglutide’s 14.9%.
But efficacy is only part of the picture.
Semaglutide has something retatrutide does not yet have: years of real-world safety data, FDA approval, and proven cardiovascular outcomes through the SELECT trial. For researchers and clinicians, that track record still matters enormously.
The honest retatrutide vs semaglutide verdict right now is this: retatrutide is the more powerful compound. Semaglutide is the more proven one. Where the research sits on that spectrum will determine which compound belongs in the protocol.
Seven more Phase 3 TRIUMPH readouts are coming in 2026. The retatrutide vs semaglutide comparison will look different again by the end of the year. Watch this space. Explore our wide range of Peptide Blends for your research needs.
By the numbers, yes. The retatrutide vs semaglutide data show retatrutide producing 28.7% average weight loss at 68 weeks in the Phase 3 TRIUMPH-4 trial. Semaglutide produced 14.9% in the STEP 1 trial over the same period. That is nearly double the outcome. However, these are separate trials with different study populations. Retatrutide also lacks the long-term cardiovascular outcomes data that semaglutide has accumulated through the SELECT trial.
In Phase 3 data, retatrutide participants on 12 mg lost an average of 28.7% of their body weight, approximately 71 pounds, over 68 weeks. Semaglutide participants in STEP 1 lost an average of 14.9% over the same period. Nearly half of retatrutide participants in TRIUMPH-4 achieved at least 25% weight reduction.
A triple agonist activates three separate hormone receptors with a single molecule. Retatrutide targets GLP-1R (appetite suppression), GIPR (insulin sensitivity and fat metabolism), and GcgR, the glucagon receptor, which increases thermogenesis and energy output. This third pathway is what separates retatrutide from both semaglutide (single agonist) and tirzepatide (dual agonist).
Retatrutide is currently in Phase 3 trials under Eli Lilly’s TRIUMPH program. Seven more readouts are expected in 2026. Based on the successful TRIUMPH-4 results, Lilly is expected to file an NDA with the FDA in late 2026. A realistic approval window is early to mid-2027, though this depends on the remaining trial data.
Dysesthesia is an abnormal skin sensation; it can feel like tingling, numbness, or burning. In TRIUMPH-4, it occurred in 8.8% of participants on the 9 mg dose and 20.9% on the 12 mg dose, compared with 0.7% in the placebo group. Most cases were mild and did not lead to discontinuation. It was not observed at significant levels in the Phase 2 trial. This is a new safety signal specific to retatrutide that researchers should monitor in protocol design.
For clinical use, retatrutide is not yet available; it remains investigational. In a crossover research context, transitioning between these compounds requires careful washout planning due to their similar mechanisms and overlapping GLP-1 activity. Researchers designing cross-compound studies should account for the differing half-lives and titration schedules of each compound.
Ignite Peptides supplies both compounds as research-grade peptides with ≥99% purity and third-party COA documentation. Same-day U.S. shipping is available. View current stock and pricing on the products page.
References

Introduction Designing a precise GLP-1 titration schedule is one of
Introduction Research on the GLP-1 mechanism is one of the

Semaglutide is one of the most studied peptides in modern
Every peptide vial in a research laboratory is labelled in